
We are aware of the concept of atomic orbitals, the mathematical probability functions indicating the presence of electrons in any regional space. The bond length is around 1.20 Å.ĭo you know that an electron can be described with the help of its wave function? Therefore, the bond angle of NO2 is around 134 degrees. There we also have a bent structure but since repulsion strength is of the order of LP-LP > LP-BP > BP-BP, the bond angle order is as below 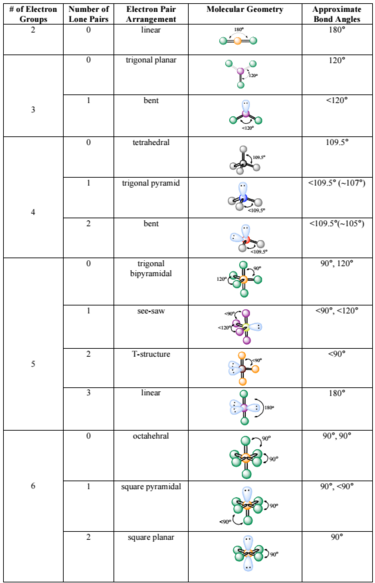
If we look at the nitrite ion NO2-, we have 2 Bond Pairs and 1 Lone pair of electrons. In NO2, we have 2 Bond Pairs and 1 lone electron. 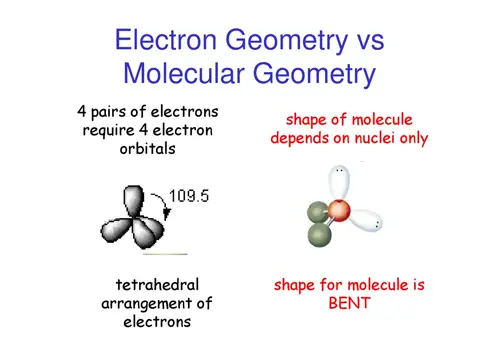
We can see that NO2 has a bent molecular geometry and the angle is around 120 degrees.īut here we have some exceptions. Here, since we have just one lone electron, Let us take it as 1. N stands for the number of atoms around the central element, we have the value of 2.Į stands for non-bonded electrons ( usually a lone pair), x stands for the number. X stands for the surrounding atoms, here we have two O atoms. In the VSEPR model, we have the AXnEx notation.Ī stands for the central atom, here we have nitrogen ( N ). VSEPR thus helps in predicting the 3D molecular geometry or shape of a molecule along with the bond angles. So, this repulsion force needs to be minimized to form a stable or balanced polyatomic molecular or ionic structure. VSEPR stands for Valence Shell Electron Pair Repulsion model.Īccording to this theory, since electrons carry like charges ( ‘-’ ), they are bound to experience repulsion and this strength of repulsion varies across bonded and lone pair electrons. This is where the role of VSEPR theory comes: It can only identify the type of bond formation but cannot elaborate on how the bond formation takes place.Lewis Structure can only talk about the arrangement of electrons but not how the electrons are shared.It can only provide a 2D representation and not a 3D one.Let us look at some of the drawbacks of Lewis Structure: Therefore, the most suitable Lewis Structure of NO2 is: Hence, the molecule is neutral and the elements are present in their least possible formal charge values. If we now find the summation of the total formal charges we get a net result of 0. Nitrogen now has 7 electrons and achieved a near octet configuration.įormal Charge of O ( in a single bond with N )įormal Charge of O ( in a double bond with N ) Now, the Nitrogen atom has only five electrons surrounding it and the total number of valence electrons has been used up already.įor the Nitrogen atom to have a near octet configuration, we are going to shift two electrons from one O atom and form a double bond. We have already made the two O atoms achieve the Neon configuration. This is known as the octet rule and is an important concept to sketch the most probable Lewis Structure of a molecule. Now, with the help of dot structures, we will surround the atoms by their valence electrons.ĭo you know that the elements present in the main groups of the periodic table have a tendency to form an eight-electron arrangement in their valence shell following noble gas configurations? This is how we have placed the nitrogen in the center surrounded by the two O atoms. Nitrogen is the least electronegative element between the two, hence N will be the central atom. Now, to find out which atom will act as a central one, we have to look at Pauling’s Electronegativity chart. Total valence electron number in NO2 = 5 + 6*2 Oxygen belongs to group 16 ( or group 6) and has an atomic number of 8, therefore a valency of 6. Nitrogen belongs to group 15( or group 5) and has an atomic number of 7, therefore has a valency of 5. Let us talk about drawing the Lewis Structure for Nitrogen Dioxide ( NO2 ).Ī molecule of nitrogen dioxide consists of one nitrogen atom and two atoms of oxygen. This is a limited theory on chemical bonding nature and electronic structure but provides a simple viewpoint towards the formation of any molecular composition. Lewis Structure is a diagrammatic representation of any given molecule with the help of the constituent atoms and the position and arrangement of electrons to form bonds and lone pairs. 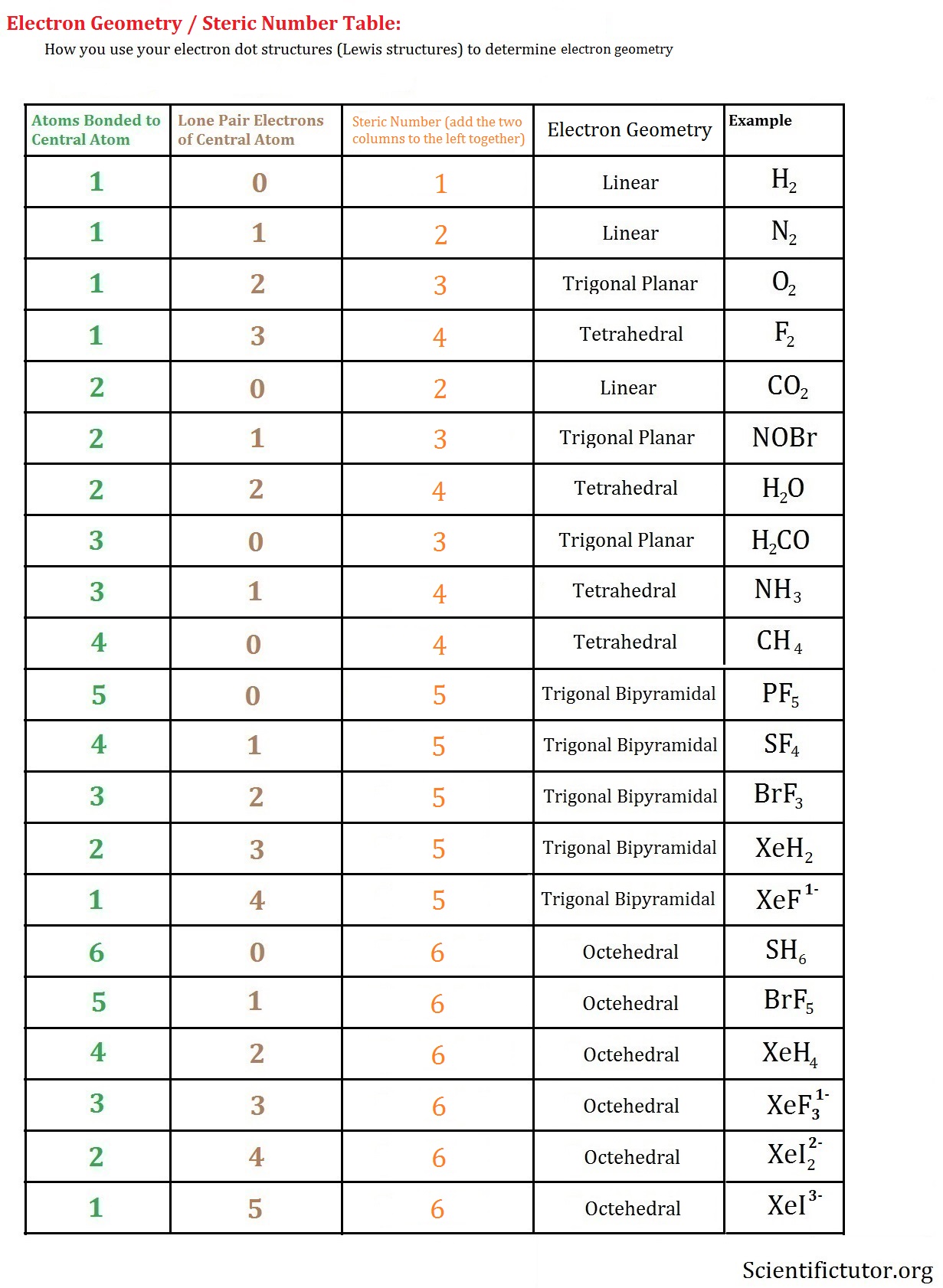
The electrons in the outermost shell which determine the valency are known as valence electrons. Inside an atom, we have the positively charged nucleus surrounded by electrons in their shells forming a negatively charged cloud.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
